T cells, a type of white blood cell, are crucial components of the immune system. These vigilant cells patrol the bloodstream, equipped with surface proteins known as receptors that detect foreign substances like viruses or bacteria. The foreign invaders also bear proteins on their surfaces, known as antigens. It’s these antigens that the T cell receptors identify and bind to, like fitting a key into a lock. This binding action triggers the T cell to destroy the foreign substance.
Interestingly, cancerous cells also possess antigens. However, it’s a rare occurrence for T cells to have the specific receptor required to bind to these cancerous antigens. This is where scientists step in and modify a person’s T cells by adding Chimeric Antigen Receptors (CARs). These CARs, functioning as new receptors, enable the T cells to recognize, bind, and annihilate cancerous cells.
The complexity arises as different cancers possess different antigens. Therefore, scientists need to tailor the treatment to suit the specific cancer. The success rate of CAR T cell therapy varies, dependent on the type of cancer being treated. A 2017 review indicated that up to 90% of patients with a certain form of leukemia saw full recovery after receiving this treatment.
CAR T cell therapy is still a relatively new approach to cancer treatment. The Food and Drug Administration (FDA) first approved it in 2017. Therefore, much remains to be learned about its efficacy. This therapy is usually considered when traditional cancer treatments, such as chemotherapy, have proven ineffective or when the cancer recurs.
In the United States, the FDA has approved four CAR T cell therapies: Breyanzi, Tecartus, Kymriah, and Yescarta. However, their use is limited to specific types of blood cancer in certain patient groups. For instance, Breyanzi is approved for adults with some forms of recurring B cell lymphoma, while Kymriah is sanctioned for patients under 25 with leukemia and adults with recurring large B cell lymphoma.
The U.S. National Library of Medicine lists over 600 ongoing clinical trials exploring the potential of CAR T cell therapy in various cancer types, including colorectal, colon, ovarian, pancreatic, liver, and lung cancers, along with acute lymphoblastic leukemia, non-Hodgkin lymphoma, acute myeloid leukemia, and multiple myeloma.
The process of CAR T cell therapy involves three steps:
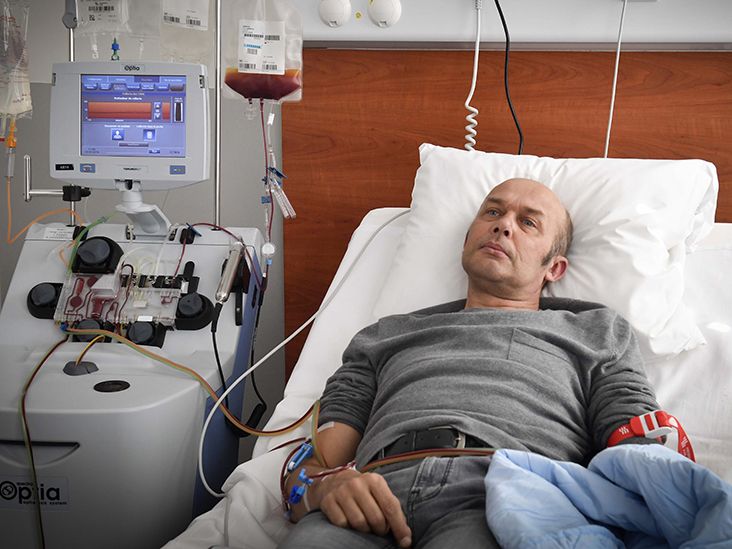
1. T cell collection: Healthcare professionals collect T cells via an intravenous (IV) line, a process that takes around 2-3 hours. Blood is drawn from the patient’s body into a machine that separates the white blood cells, which include the T cells. The remaining blood is then returned to the body.
2. CAR T cell production: T cells are separated from other white cells and sent to a laboratory, where scientists add CARs to the cells, creating CAR T cells. These modified cells are then allowed to multiply before being reinfused into the patient. This step can take a few weeks.
3. CAR T cell infusion: The CAR T cells are reintroduced into the patient’s bloodstream via an IV line. Some patients may require chemotherapy to prepare their immune system for the new CAR T cells.
However, CAR T cell therapy can cause side effects, the most common being cytokine release syndrome (CRS). CRS occurs when there’s an overproduction of cytokines, chemicals that aid T cells, triggered by the introduction of CAR T cells. CRS symptoms range from mild—like nausea, fatigue, headache, chills, and fever—to severe, including low blood pressure, rapid heartbeat, cardiac arrest, heart failure, hypoxia, impaired kidney function, and multiple organ failure. Severe CRS can also cause neurological issues, such as language impairment, confusion, delirium, involuntary muscle twitching, hallucinations, unresponsiveness, and seizures. Severe CRS is highly dangerous and requires immediate intensive care treatment. While most symptoms are reversible, CRS can occasionally be fatal.