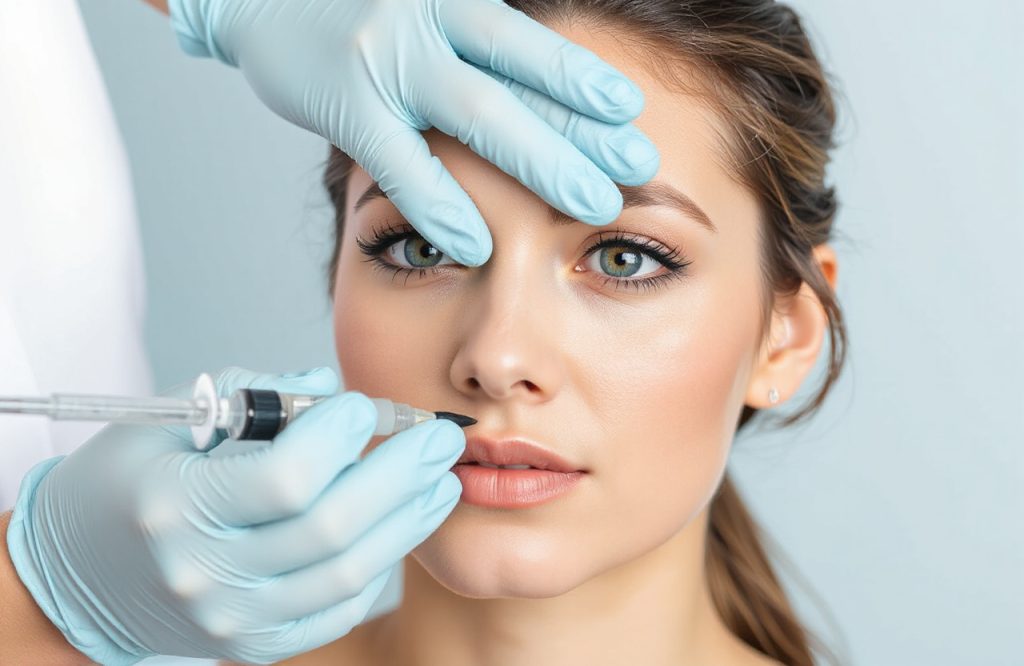
Introduction to Hyaluronic Acid Fillers
Standard for Non-Surgical Corrections
Hyaluronic Acid (HA) fillers are widely recognized as the standard treatment for non-surgical corrections of nasolabial folds and crow’s feet. However, clinicians continually seek innovative options to enhance patient experience and outcomes.
Evaluation of New HA Fillers
Study Overview
A comprehensive evaluation of new HA fillers infused with lidocaine has shown results that are comparable to existing formulations. Hyaluronic acid is the most commonly used facial filler to address significant signs of aging, including deepened nasolabial folds and wrinkles around the eyes.
Mechanism and Composition
Viscos HA is injected into the dermis to fill in furrows and maintain facial shape. Its unique composition minimizes adverse reactions and side effects, making it a safe non-surgical option for combating facial aging.
Limitations of Current HA Fillers
Pain Management Challenges
Despite the popularity of HA fillers, they are not without drawbacks. Subdermal injections often cause discomfort. To address this issue, newer formulations have been developed that combine the benefits of HA with the anesthetic properties of lidocaine.
Comparative Study of Fillers
Research Methodology
To assess the potential of these new fillers, dermatologists conducted a rigorous study involving over 70 patients across multiple centers in Europe. Patients received filler injections targeting either nasolabial folds, crow’s feet, or both, with follow-ups to evaluate efficacy and tolerability.
Blinded Study Design
A split-face methodology was employed, where one side of the face was treated with a standard HA filler while the other side received the experimental formulation. This blinding of both patients and physicians eliminated potential biases in the results.
Results and Future Directions
Findings and Efficacy
Patients were monitored at 30 and 45 days post-treatment. Objective evaluations indicated that the results achieved with both established and newer formulations were comparable in terms of efficacy and tolerability.
Next Steps in Research
Encouraged by these findings, further studies are planned to assess the results at longer intervals, specifically at 90 and 180 days post-treatment. If these newer HA formulations demonstrate equivalent efficacy, they may eventually replace current options in the market, offering a less painful alternative for addressing signs of aging.
Conclusion
The introduction of HA fillers infused with lidocaine represents a promising advancement in the field of aesthetic medicine, potentially transforming the patient experience and treatment outcomes in facial rejuvenation.
Author Information
Written By: Jay Martin, M.D.